პროდუქტის დასახელება:მეთილ ეთილ კეტონი
მოლეკულური ფორმატი:C4H8O
CAS ნომერი:78-93-3
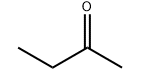
პროდუქტის მოლეკულური სტრუქტურა:
სპეციფიკაცია:
| ნივთი | ერთეული | ღირებულება |
| სიწმინდე | % | 99.8 წთ |
| ფერი | APHA | 8 მაქსიმუმი |
| მჟავიანობის მაჩვენებელი (აცეტატის მჟავას სახით) | % | 0.002 მაქს. |
| ტენიანობა | % | 0.03 მაქს. |
| გარეგნობა | - | უფერო სითხე |
ქიმიური თვისებები:
მეთილეთილკეტონი მგრძნობიარეა სხვადასხვა რეაქციების მიმართ მისი კარბონილის ჯგუფისა და კარბონილის ჯგუფთან მიმდებარე აქტიური წყალბადის გამო. კონდენსაცია ხდება მარილმჟავით ან ნატრიუმის ჰიდროქსიდით გაცხელებისას 3,4-დიმეთილ-3-ჰექსენ-2-ონის ან 3-მეთილ-3-ჰეპტენ-5-ონის მისაღებად. მზის შუქზე ხანგრძლივი ზემოქმედებისას წარმოიქმნება ეთანი, ძმარმჟავა და კონდენსაციის პროდუქტები. აზოტმჟავით დაჟანგვისას წარმოიქმნება დიაცეტილი. ძლიერი დამჟანგავი საშუალებებით, როგორიცაა ქრომის მჟავა, დაჟანგვისას წარმოიქმნება ძმარმჟავა. ბუტანონი შედარებით სტაბილურია სითბოს მიმართ და მაღალ ტემპერატურაზე თერმული დაშლისას წარმოიქმნება ენონი ან მეთილენონი. ალიფატურ ან არომატულ ალდეჰიდებთან კონდენსაციისას წარმოიქმნება მაღალი მოლეკულური წონის კეტონები, ციკლური ნაერთები, კეტონური კონდენსაცია და ფისები. მაგალითად, ფორმალდეჰიდთან კონდენსაცია ნატრიუმის ჰიდროქსიდის თანაობისას თავდაპირველად წარმოქმნის 2-მეთილ-1-ბუტანოლ-3-ონს, რასაც მოჰყვება დეჰიდრატაცია მეტაკრილატონამდე.
რეზინიზაცია ხდება მზის ან ულტრაიისფერი სინათლის ზემოქმედებისას. ფენოლთან კონდენსაციის შედეგად მიიღება 2,2-ბის(4-ჰიდროქსიფენილ)ბუტანი. ფუძე კატალიზატორის თანაობისას რეაგირებს ალიფატურ ეთერებთან β-დიკეტონების წარმოსაქმნელად. მჟავე ანჰიდრიდთან აცილირების შედეგად მჟავე კატალიზატორის თანაობისას წარმოიქმნება β-დიკეტონები. რეაგირებს წყალბადის ციანიდთან ციანოჰიდრინის წარმოსაქმნელად. რეაგირებს ამიაკთან კეტოპიპერიდინის წარმოებულების წარმოსაქმნელად. ბუტანონის α-წყალბადის ატომი ადვილად იცვლება ჰალოგენებით სხვადასხვა ჰალოგენიზებული კეტონების წარმოსაქმნელად, როგორიცაა 3-ქლორ-2-ბუტანონი ქლორთან ურთიერთქმედებით. 2,4-დინიტროფენილჰიდრაზინთან ურთიერთქმედებისას წარმოიქმნება ყვითელი 2,4-დინიტროფენილჰიდრაზონი.
განაცხადი:
მეთილ ეთილ კეტონი (2-ბუტანონი, ეთილ მეთილ კეტონი, მეთილ აცეტონი) შედარებით დაბალი ტოქსიკურობის ორგანული გამხსნელია, რომელიც მრავალ დანიშნულებაში გვხვდება. იგი გამოიყენება სამრეწველო და კომერციულ პროდუქტებში, როგორც წებოვანი ნივთიერებების, საღებავებისა და საწმენდი საშუალებების გამხსნელი და ცვილის მოსაშორებელი გამხსნელი. ზოგიერთი საკვების ბუნებრივი კომპონენტი, მეთილ ეთილ კეტონი, შეიძლება გარემოში გამოიყოფა ვულკანებისა და ტყის ხანძრების შედეგად. იგი გამოიყენება უკვამლო ფხვნილისა და უფერო სინთეზური ფისების წარმოებაში, როგორც გამხსნელი და ზედაპირის საფარი. იგი ასევე გამოიყენება როგორც არომატიზატორი საკვებში.
MEK გამოიყენება გამხსნელად სხვადასხვა საფარის სისტემებისთვის, მაგალითად, ვინილის, წებოვანი მასალების, ნიტროცელულოზის და აკრილის საფარებისთვის. იგი გამოიყენება საღებავის მოსაშორებელ საშუალებებში, ლაქებში, სპრეი საღებავებში, დალუქვის საშუალებებში, წებოებში, მაგნიტურ ლენტებში, საბეჭდ მელნებში, ფისებში, როზინებში, საწმენდ ხსნარებში და პოლიმერიზაციისთვის. იგი გვხვდება სხვა სამომხმარებლო პროდუქტებში, მაგალითად, საყოფაცხოვრებო და ჰობის ცემენტებში და ხის შემავსებელ პროდუქტებში. MEK გამოიყენება საპოხი ზეთების დეცილაციისთვის, ლითონების ცხიმის მოცილებისთვის, სინთეტიკური ტყავის, გამჭვირვალე ქაღალდისა და ალუმინის ფოლგის წარმოებაში და როგორც ქიმიური შუალედური პროდუქტი და კატალიზატორი. ის არის ექსტრაქციის გამხსნელი საკვები პროდუქტებისა და საკვები ინგრედიენტების დამუშავებაში. MEK ასევე შეიძლება გამოყენებულ იქნას ქირურგიული და სტომატოლოგიური აღჭურვილობის სტერილიზაციისთვის.
წარმოების გარდა, MEK-ის გარემო წყაროებია რეაქტიული და შიგაწვის ძრავების გამონაბოლქვი და სამრეწველო საქმიანობა, როგორიცაა ნახშირის გაზიფიკაცია. ის მნიშვნელოვანი რაოდენობით გვხვდება თამბაქოს კვამლში. MEK ბიოლოგიურად წარმოიქმნება და იდენტიფიცირებულია, როგორც მიკრობული მეტაბოლიზმის პროდუქტი. ის ასევე გვხვდება მცენარეებში, მწერების ფერომონებსა და ცხოველთა ქსოვილებში და MEK, სავარაუდოდ, ნორმალური ძუძუმწოვრების მეტაბოლიზმის მცირე პროდუქტია. ის სტაბილურია ჩვეულებრივ პირობებში, მაგრამ ხანგრძლივი შენახვისას შეიძლება წარმოქმნას პეროქსიდები; ისინი შეიძლება ასაფეთქებელი იყოს.
პროდუქტების კატეგორიები
-

ტელეფონი
-

ელ. ფოსტა
-

WhatsApp
-

ზედა













